Apoptosis Signaling Laboratory

Senior lecturer: Dr. Zsolt Sarang, Dr. Krisztina Köröskényi
Assistant lecturers: Dr. Éva Garabuczi, Dr. Beáta Kiss, Dr. Zsófia Buda
PhD students: Éva Fige, Nour Al Zayed, Nastaran Tarban, Lulu Alsheikh Hussein, László Sós
Technician: Evelin Erdős

CONTACTS
| Office address: |
Department of Biochemistry and Molecular Biology |
| University of Debrecen | |
| Egyetem tér 1 | |
| H-4010 Debrecen | |
| FAX: | +36 52 314-989 |
| Name: | Dr. Zsuzsanna Szondy |
| E-Mail: | szondy@med.unideb.hu |
| Phone: | +36 52 416-432 -64134 |
| Name: | Dr. Krisztina Köröskényi, Dr. Zsolt Sarang, Dr. Éva Garabuczi, Dr. Beáta Kiss, Dr. Zsófia Budai |
| E-Mail: | kkriszti@med.unideb.hu sarang@med.unideb.hu gevi@med.unideb.hu kissbea@med.unideb.hu budai.zsofia@med.unideb.hu |
| Phone: | +36 52 416-432 -64789 |
| Name: |
Éva Fige, Nour Al Zayed, Nastaran Tarban, Lulu Alsheikh Hussein, László Sós Evelin Erdős |
| E-Mail: | |
| Phone: | +36 52 416-432 -65899 |
RESEARCH TOPICS
Every day one billion cells die and are replaced by new cells in a human body. The death is part of our healthy tissue cell turnover, but cells can die also after infection or injury. The dead cells are cleared mostly by professional phagocytes which however not only clear and degrade the dead cells, but they also decide whether the dead cells were healthy and silent clearance is required, or the cell was an infected, necrotic or cancer cell, and then inflammation has to be triggered. Inflammatory macrophages clear dead cells, initiate an immune response that kills and removes all the dangerous cells, but then they are converted to healing macrophages that silence the immune response and orchestrate the tissue repair. Increasing evidence indicate that improper phagocytosis and immune silencing leads to chronic inflammatory and autoimmune diseases. The interest of our group is to find the mechanisms through which dead cells are recognized and cleared, and also the mechanisms through which the inflammation is silenced and tissues are repaired with the long term goal of influencing these processes in chronic inflammatory diseases.
SELECTED PUBLICATIONS:
Sarang Z, Sághy T, Budai Z, Újlaky-Nagy L, Bedekovics J, Beke L, Méhes G, Nagy G, Rühl R, Moise AR, Palczewski K, Szondy Z. (2019) Retinol Saturase Knock Out Mice are Characterized with Impaired Clearance of Apoptotic Cells and Develop Mild Autoimmunity. Biomolecules 9:737.
Farkas Z, Petric M, Liu X, Herit F, Rajnavölgyi É, Szondy Z, Budai Z, Orbán TI, Sándor S, Mehta A, Bajtay Z, Kovács T, Jung SY, Afaq Shakir M, Qin J, Zhou Z, Niedergang F, Boissan M, Takács-Vellai K. ( 2019) The nucleoside diphosphate kinase NDK-1/NME1 promotes phagocytosis in concert with DYN-1/Dynamin. FASEB J. 33(10):11606-11614.
Sághy T, Köröskényi K, Hegedűs K, Antal M, Bankó C, Bacsó Z, Papp A, Stienstra R, Szondy Z. (2019) Loss of transglutaminase 2 sensitizes for diet-induced obesity-related inflammation and insulin resistance due to enhanced macrophage c-Src signaling. Cell Death Dis. 10(6):439.
Budai Z, Ujlaky-Nagy L, Kis GN, Antal M, Bankó C, Bacsó Z, Szondy Z, Sarang Z. (2019) Macrophages engulf apoptotic and primary necrotic thymocytes through similar phosphatidylserine- dependent mechanisms. FEBS Open Bio. 9(3):446-456.
Köröskényi K, Joós G, Szondy Z. (2017) Adenosine in the Thymus. Front Pharmacol. 8:932.
Szondy Z, Sarang Z, Kiss B, Garabuczi É, Köröskényi K. Anti-inflammatory Mechanisms Triggered by Apoptotic Cells during Their Clearance.(2017) Front Immunol. 8:909.
Köröskényi K, Kiss B, Szondy Z (2016) Adenosine A2A receptor signaling attenuates LPS-induced pro-inflammatory cytokine formation of mouse macrophages by inducing the expression of DUSP1. BBA Mol. Cell Res. 1863(7 Pt A):1461-71.
Garabuczi E, Sarang Z, Szondy Z (2015) Glucocorticoids enhance prolonged clearance of apoptotic cells by upregulating liver X receptor, peroxisome proliferator-activated receptor-δ and UCP2.BBA Mol. Cell Res. 1853: 573–582
Szondy Z, Garabuczi E, Joós G, Tsay GJ, Sarang Z (2014) Impaired clearance of apoptotic cells in chronic inflammatory diseases: therapeutic implications. Front Immunol. 5: 354.
Duró E, Pallai A, Köröskényi K, Sarang Z, Szondy Z.(2014) Adenosine A3 receptors negatively regulate the engulfment-dependent apoptotic cell suppression of inflammation. Immunol Lett. 162(2 Pt B): 292-301.
Yen JH, Yang DJ, Chen MC, Yi-Ying W, Hsieh YF, Cheng YM, Huang WN, Szondy Z, Tsay GJ (2014) Daidzein enhances efferocytosis via transglutaminase 2 and augmentation of Rac1 activity. Mol. Immunol. 60: 135-142.
Sarang Z, Joós G, Garabuczi E, Rühl R, Gregory CD, Szondy Z (2014) Macrophages Engulfing Apoptotic Cells Produce Non-classical Retinoids to Enhance Their Phagocytic Capacity. J. Immunol. 192: 5730-5738.
Sarang Z, Garabuczi E, Joós G, Kiss B, Tóth K, Rühl R, Szondy Z (2013) Macrophages engulfing apoptotic thymocytes produce retinoids to regulate selection, removal and replacement of double positive thymocytes. Immunobiology 13: 122-128.
Szondy Z, Garabuczi E, Tóth K, Kiss B & Köröskényi K (2012) Thymocyte death by neglect: contribution by engulfing macrophages. Eur J Immunol 42:1662-1667.
Szondy Z, Korponay-Szabó I, Király R, Fésüs L. Transglutaminase 2 dysfunctions in the development of autoimmune disorders: celiac disease and TG2-/- mouse. (2011) Adv Enzymol Relat Areas Mol Biol. 78: 295-345.
Köröskényi K, Duró E, Pallai A, Sarang Z, Kloor D, Ucker D, Ledent CA, Chawla A, Castrillo A, Fésüs L, Szondy Z (2011) Involvement of Adenosine A2A Receptors in Engulfment-Dependent Apoptotic Cell Suppression of Inflammation. J Immunol 186: 7144-7155.
Tóth K, Sarang Z, Brázda P, Ghyselinck N, Chambon P, Fésüs L, Szondy Z. (2011) Retinoids enhance glucocorticoid-induced apoptosis of T cells by facilitating glucocorticoid receptor- mediated transcription. Cell Death Differ 18:783-792.
Tóth B, Garabuczi E, Sarang Z, Vereb G, Vámosi G, Aeschlimann D, Blaskó B, BécsiB, Erdődi F, Lacy-Hulbert A, Zhang A, Falasca L, Balajthy Z, Birge R, Melino G, Fésüs L, Szondy Z (2009) Transglutaminase 2 is needed for the formation of an efficient phagocyte portal in macrophages engulfing apoptotic cells. J Immunol 182: 2084-2092.
Szondy Z, Sarang Z, Molnár P, Németh T, Piacentini M, Mastroberardino PG, FalascaL, Aeschlimann D, Szegezdi E, Lakos G, Kovács J, Rajnavölgyi E, Birckbichler J, Melino G, Fésüs L (2003) TGASE2-/- mice reveal a phagocytosis-associated crosstalk between macrophages and apoptotic cells. Proc Natl Acad Sci USA 100:7812-7817.
Pictures from the laboratory




Pictures from papers


Macrophages engulfing a dying adipocyte. Macrophages were stained green with blue nucleus, while the fat content of dying adipocytes is red. Dying adipocytes pack their lipid content into small vesicles (seen like small red balls) that are taken up first by the green macrophages. When the red and green color overlap in them macrophages become yellow indicating fat in them as well. Cytoplasmic content of the dying adipocytes form colorless (fatless) apoptotic bodies that are small enough to be engulfed by the macrophages also.
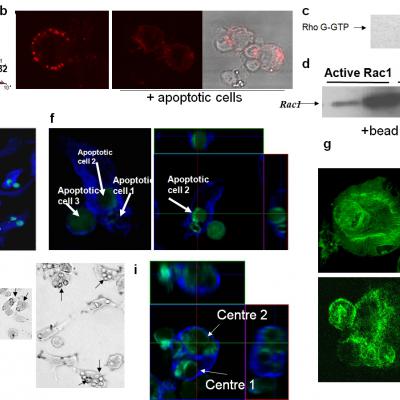
Macrophages engulfing apoptotic thymocytes.
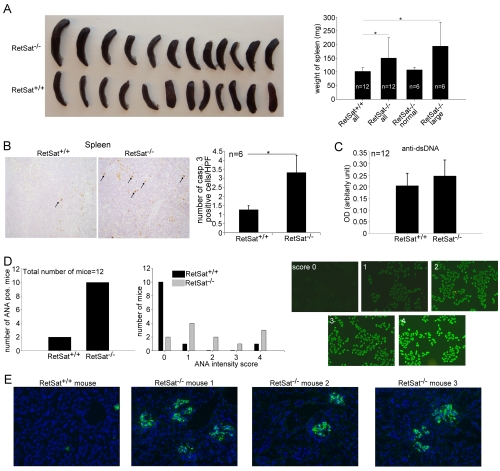
If removal of dead cells is impaired , a big proportion of mice develop autoimmunity. Autoimmunity is characterized with an overwhelmed immune response through which the immune system attacks our own body. These mice are characterized by (A) enlarged spleen, ((B) dead cell accumulation in tissues, (D) antibodies against the components of the nuclei of our cells and (E) autoantibody complexes (green) deposited into the kidney causing kidney failure. The same things occur in the human body.
News
New Ph.D. defences in 2020
Zsófia Budai
Tibor Sághy